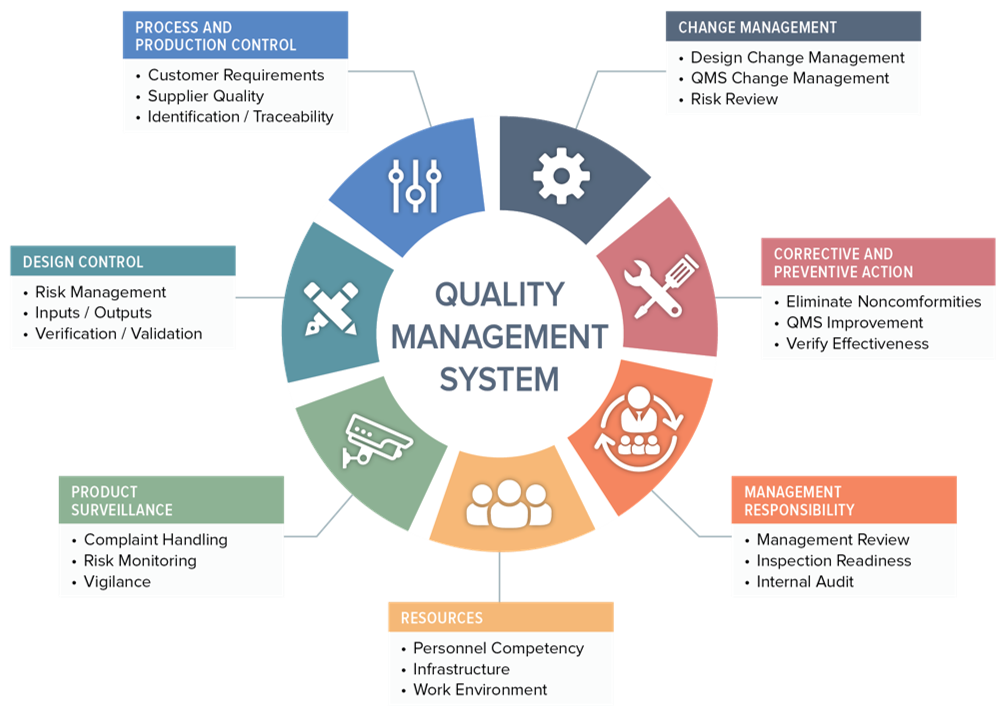
Medical Device Management Review . organizations that place medical devices on the market under their own name (often referred to as the ‘legal. this guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. the overall objective of the hse’s medical device equipment management policy is to provide an organisation wide framework for. the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. Discover key strategies for compliance and patient safety, from rigorous staff training to digital qms. navigating medical device management can be complex. this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to where the device is sold. To ensure consistent compliance with customer and regulatory requirements. the key aims of the quality management review for medical devices are:
from www.orielstat.com
this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to where the device is sold. To ensure consistent compliance with customer and regulatory requirements. the key aims of the quality management review for medical devices are: this guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. navigating medical device management can be complex. organizations that place medical devices on the market under their own name (often referred to as the ‘legal. Discover key strategies for compliance and patient safety, from rigorous staff training to digital qms. the overall objective of the hse’s medical device equipment management policy is to provide an organisation wide framework for.
Medical Device QMS 101 What It Is, Where It’s Required, and Key
Medical Device Management Review organizations that place medical devices on the market under their own name (often referred to as the ‘legal. navigating medical device management can be complex. this guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. organizations that place medical devices on the market under their own name (often referred to as the ‘legal. To ensure consistent compliance with customer and regulatory requirements. the overall objective of the hse’s medical device equipment management policy is to provide an organisation wide framework for. the key aims of the quality management review for medical devices are: the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to where the device is sold. Discover key strategies for compliance and patient safety, from rigorous staff training to digital qms.
From www.cognidox.com
4 ways to build a medical device quality management system Medical Device Management Review the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. organizations that place medical devices on the market under their own name (often referred to as the ‘legal. this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to. Medical Device Management Review.
From www.syscreations.com
Medical Device Integration Solutions With EMR, EHR, LIS, HIS Medical Device Management Review the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. the key aims of the quality management review for medical devices are: navigating medical device management can be complex. To ensure consistent compliance with customer and regulatory requirements. organizations that place medical devices on the market under. Medical Device Management Review.
From edu.svet.gob.gt
ISO Standards For Medical Devices Ultimate List Overview Medical Device Management Review the overall objective of the hse’s medical device equipment management policy is to provide an organisation wide framework for. navigating medical device management can be complex. organizations that place medical devices on the market under their own name (often referred to as the ‘legal. this is to ensure that the quality system satisfies all applicable requirements. Medical Device Management Review.
From www.greenlight.guru
The Ultimate Guide To Design Controls For Medical Device Companies Medical Device Management Review the key aims of the quality management review for medical devices are: the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. the overall objective of the hse’s medical device equipment management policy is to provide an organisation wide framework for. navigating medical device management can be. Medical Device Management Review.
From www.joharidigital.com
Medical Device Development Design Control Johari Digital Healthcare Ltd. Medical Device Management Review To ensure consistent compliance with customer and regulatory requirements. this guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. the key aims of the quality management review for medical devices are: organizations that place medical devices on the market under their own name (often referred to as the ‘legal.. Medical Device Management Review.
From www.researchgate.net
(PDF) Health technology assessment as a tool for medical devices Medical Device Management Review Discover key strategies for compliance and patient safety, from rigorous staff training to digital qms. navigating medical device management can be complex. this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to where the device is sold. To ensure consistent compliance with customer and regulatory requirements. the. Medical Device Management Review.
From www.orielstat.com
Medical Device QMS 101 What It Is, Where It’s Required, and Key Medical Device Management Review Discover key strategies for compliance and patient safety, from rigorous staff training to digital qms. organizations that place medical devices on the market under their own name (often referred to as the ‘legal. this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to where the device is sold.. Medical Device Management Review.
From www.researchgate.net
(PDF) Documenting Medical Device Risk Management through the Risk Medical Device Management Review the overall objective of the hse’s medical device equipment management policy is to provide an organisation wide framework for. this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to where the device is sold. To ensure consistent compliance with customer and regulatory requirements. organizations that place medical. Medical Device Management Review.
From studylib.net
Medical Devices Management Policy V7.0 Medical Device Management Review To ensure consistent compliance with customer and regulatory requirements. Discover key strategies for compliance and patient safety, from rigorous staff training to digital qms. this guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of. Medical Device Management Review.
From pharmait.dk
Medical Device Quality Management System processes Pharma IT Medical Device Management Review this guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. the key aims of the quality management review for medical devices are: navigating medical device management can be complex.. Medical Device Management Review.
From www.researchgate.net
Device Connectivity Architecture Manager MDD. The medical device Medical Device Management Review navigating medical device management can be complex. the key aims of the quality management review for medical devices are: this guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. organizations that place medical devices on the market under their own name (often referred to as the ‘legal. . Medical Device Management Review.
From www.modernrequirements.com
Facilitate the Medical Device Design Controls Modern Requirements Medical Device Management Review Discover key strategies for compliance and patient safety, from rigorous staff training to digital qms. the key aims of the quality management review for medical devices are: To ensure consistent compliance with customer and regulatory requirements. this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to where the. Medical Device Management Review.
From www.walmart.com
Medical Device Manager Medical Device Management Review organizations that place medical devices on the market under their own name (often referred to as the ‘legal. the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. this guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. Discover key. Medical Device Management Review.
From www.slideshare.net
6 Ways to Improve Your Medical Device Management Medical Device Management Review this guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. navigating medical device management can be complex. the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. the key aims of the quality management review for medical devices are:. Medical Device Management Review.
From kvalito.ch
Risk Management for Medical Devices ISO 149712019 Kvalito Medical Device Management Review organizations that place medical devices on the market under their own name (often referred to as the ‘legal. the overall objective of the hse’s medical device equipment management policy is to provide an organisation wide framework for. the key aims of the quality management review for medical devices are: Discover key strategies for compliance and patient safety,. Medical Device Management Review.
From templates.rjuuc.edu.np
Medical Device Design Review Template Medical Device Management Review this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to where the device is sold. the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. To ensure consistent compliance with customer and regulatory requirements. the key aims of. Medical Device Management Review.
From financesonline.com
Best Medical Practice Management Software in 2024 Medical Device Management Review organizations that place medical devices on the market under their own name (often referred to as the ‘legal. the key aims of the quality management review for medical devices are: the purpose of the iso 13485 management review is to evaluate the effectiveness and suitability of an organization’s quality. Discover key strategies for compliance and patient safety,. Medical Device Management Review.
From spyro-soft.com
Do you need a certified Quality Management System to introduce a Medical Device Management Review this is to ensure that the quality system satisfies all applicable requirements of medical devices, which are mainly related to where the device is sold. navigating medical device management can be complex. the key aims of the quality management review for medical devices are: the overall objective of the hse’s medical device equipment management policy is. Medical Device Management Review.